The formation history of carboxylic acids in different environments is still largely unknown. In this study, the pathway of one-electron oxidation was observed at hydrothermal conditions through experiments with iron oxide minerals. This pathway provides a new explanation for the formation of carboxylic acids and how carboxylic acids can be altered in geologic environments. Carboxylic acid structures can evolve through a complex network of reactions that occurs only in the presence of minerals. The formation of reaction cycles and secondary pathways could be part of the pre-biotic history of our planet and be a framework for primitive metabolism.
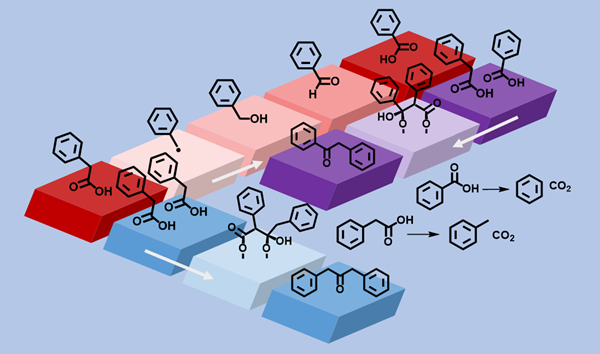
The fate of organic compounds in the geologic record is likely complex and evolving with time, following different pathways based on what a compound is subjected to while existing in a specific space. Mapping potential pathways through experiments provide information for how and when those pathways can occur. Multiple fates can await a single type of compound, taking progressive mechanistic steps to form new products. Some intermediate forms of an organic compound will be difficult to observe experimentally, while others will become reagents in a new sequence of reactions. Reaction pathways for an organic compound can either require the involvement of a mineral surface or be possible in water alone [Figure 1].
Understanding the complexity possible within the geologic environment from non-biological (abiotic) reactions allows speculation regarding what was possible on the early Earth or interpretation of signals from other planets in the search for extraterrestrial life. Hot aqueous environments such as hydrothermal vents and subduction zones at the bottom of the ocean offer promising environments for interesting chemistry to occur. Detailed hydrothermal experimental studies into the specific pathways possible for organic compounds in the presence of minerals can expand our understanding, challenging our assumptions about the possible fates that await a single organic compound within a geologic context.
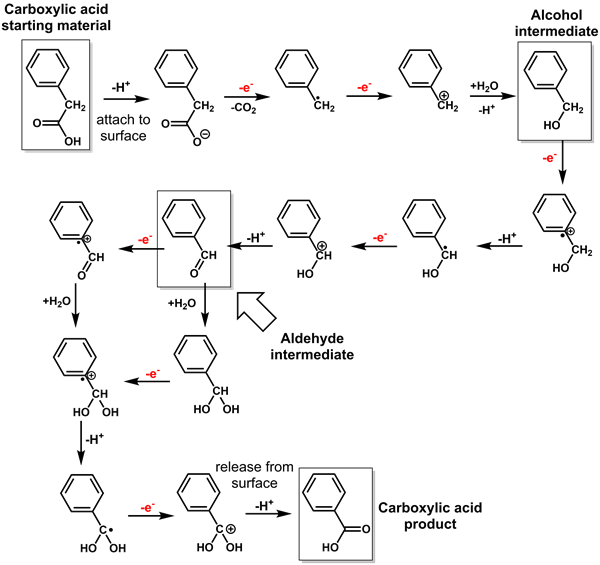
In addition to being a part of biology through essential metabolic cycles, carboxylic acids are found abundantly in geologic systems. Within the Murchison meteorite, carboxylic acids are one of the most abundant organic compound groups measured. Still, the origin of the many diverse forms and structures of these compounds has remained unexplained. Previously, author Kristin N. Johnson-Finn showed a pathway for the consumption of carboxylic acids in the formation of ketones in the presence of four different metal oxide minerals (i.e. magnetite, hematite, corundum, and spinel) at hydrothermal conditions. The current study expands on that work, explaining and demonstrating a new reaction pathway for forming smaller carboxylic acids from larger carboxylic acids: one-electron oxidation. This pathway has not been previously considered or described in the geochemical literature as a method for carboxylic acid formation. This new pathway was observed in the presence of magnetite and hematite (redox-active, iron oxide minerals). The research team propose that the carboxylic acid formation pathway requires the availability of a redox-active metal oxide because this pathway was not observed in the presence of corundum or spinel minerals in the previous study. Both corundum and spinel are non-redox active and would be considered insulator materials, unable to store or transport an electron. The formation pathway of a short-chain carboxylic from the longer carboxylic acid starting material is progressive one-electron oxidation. Intermediate alcohol and aldehyde forms are created and rapidly consumed during the reaction mechanism to form the final carboxylic acid [Figure 2].
Once this new reaction pathway becomes available, the complexity of the entire system builds. In water, there is only one fate of reaction. With a non-redox active metal oxide, there become two primary fates of reaction and a few secondary pathways. With the redox-active metal oxide, there are three starting reaction pathways and a growing number of complexity-building secondary pathways. Some reactions appear to be reversible, while others irreversibly form hydrocarbon products unable to rejoin the complex network that evolves over time. A reaction of note is the coupling of both forms of carboxylic acids to create a ketone product that was otherwise unable to form without the existence of the shorter chain carboxylic acid. Beginning with only one carboxylic acid starting material and one iron oxide mineral (either magnetite or hematite), the result is a complex array of products either occurring on the mineral surface or the result of redox reactions involving the loss of electrons [Figure 3].
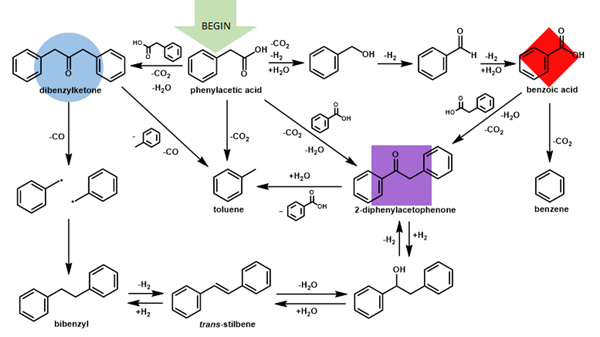
Complexity builds from the addition of chemical properties supplied by the mineral and the available structural components of the organic compound. In this study, the research team tested the observed reaction network's universality through experiments starting with a more complex carboxylic acid. The result of those experiments was the formation of a similar reaction network and the development of many more reaction products. Universality appears possible across classes of compounds, but additional products can also become available based on the structure of the starting organic compound. The number of products detected made it too difficult to develop an exact reaction web diagram. Still, it was evident that adding a one-carbon chain changed the reaction rate and the available secondary products significantly in the presence of magnetite.
Complex, interrelated systems appear to develop from simple starting conditions. Coupled with other geologic environments, could the first metabolic cycle for carboxylic acids emerge?
| Journal | ACS Earth and Space Chemistry |
| Tile of the paper | Hydrothermal One-Electron Oxidation of Carboxylic Acids in the Presence of Iron Oxide Minerals |
| Authors | Kristin N. Johnson-Finna,b*, Lynda B. Williamsc, Ian R. Gouldb, Hilairy E. Hartnettb,c, Everett L. Shockb,c |
| Affiliations | (a) Earth-Life Science Institute, Tokyo Institute of Technology, Tokyo, Tokyo 152-8550, Japan. (b) School of Molecular Sciences, Arizona State University, Tempe, AZ, 85287, United States (c) School of Earth & Space Exploration, Arizona State University, Tempe, AZ, 85287, United States |
| DOI | 10.1021/acsearthspacechem.1c00139 |
| Online published date | September 17, 2021 |
