Abstract:
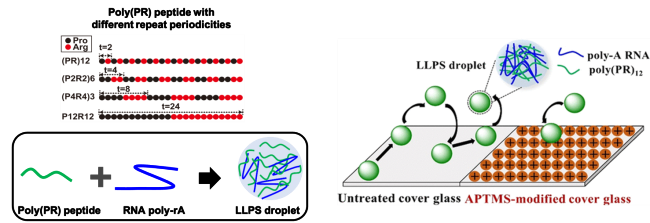
Liquid-liquid phase separation (LLPS) of proteins and RNA emerges as the ubiquitous phenomenon related to the condensed organization of living cells and cellular functions. Recently, Arg-rich dipeptide repeat proteins (DPRs), especially poly(PR), encoded by a hexanucleotide expansion in C9ORF72 gene are responsible for the neurodegeneration in amyotrophic lateral sclerosis (ALS). Most importantly, these poly(PR) dipeptide repeats are known to undergo LLPS and affect multiple biological processes. Moreover, these LLPS droplets possess liquid-like features, such as spherical shape, fusion and fission, wetting surface, etc. However, there still exists several mysteries for these droplets research. Among them, one is the intermolecular interaction for LLPS formation based on the structural distribution of positively-charged Arg and rigid Pro residues in polypeptide; the other is the interaction between LLPS interface with outer medium, which could affect LLPS uptake process for surrounding biomolecules. This work conducted a series of computations and experiments and found the primary sequence of dipeptides caused unique charge distributions and molecular rigidity, which correlated to the biding energy to RNA. Inserted Pro residues functioned as spacers and provided multivalency to from LLPS; the alternate Arg structure could efficiently trap proteins with acidic stretches (including NPM1 protein) by electrostatic interactions. In addition, we utilized the single-particle tracking method to observe the dynamic behaviours of LLPS droplets at two substrates (one is untreated cover glass and the other is chemically-modified cover glass with amine groups). We revealed there were mainly fix and diffusion modes for droplets diffusing at the solid/liquid interface: most LLPS droplets exhibited dynamic diffusion mode on the untreated cover glass surface while most LLPS droplets were fixed on the chemically-modified cover glass surface with amine groups. In summary, the research will provide a fundamental explanation for understanding LLPS formation and dynamics, which may be helpful to explain the impedance of functions of membrane-less organelles including nucleoli and ALS disease pathology. Moreover, we also introduced an improved method to control LLPS uptake process for surrounding biomolecules through the interaction between droplets surface and the outer medium, which is also meaningful for ALS disease curation in the near future.
Speaker: Dr. Chen Chen, Earth-Life Science Institute, Tokyo Institute of Technology.

Speaker bio:
Chen Chen is currently a postdoctoral researcher in Earth-Life Science Institute at Tokyo Institute of Technology. He received his Ph.D. degree in Materials Science and Engineering at Tokyo Institute of Technology this year. His research interest is biomolecular self-assembly and biological liquid-liquid phase separation (LLPS). Now, he is working with Prof. Tony Z. Jia to explore the LLPS in the context of origins of life and protocells.
Host: Tony Z. Jia, ELSI.
Date: Fri, 6 August, 10:30-11:30 JST
Venue: Online
